




1 / 5










Wound Protectors/Retractors are for disposable use in stabilizing and protecting wound incision in surgery.
Maximum exposure of a minimum incision; Maximum surgical view; Broaden surgical access.
Prevention of infection; Minimize tissue damage; Minimize bleeding of wound incision.
Designed to save critical surgical time during procedures.
Available in multiple sizes to meet different needs of surgeons.



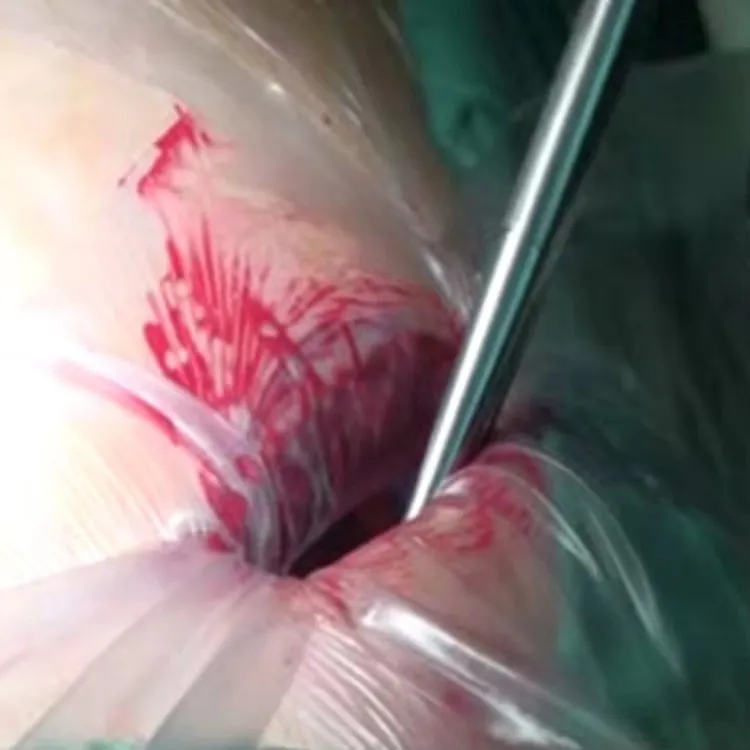
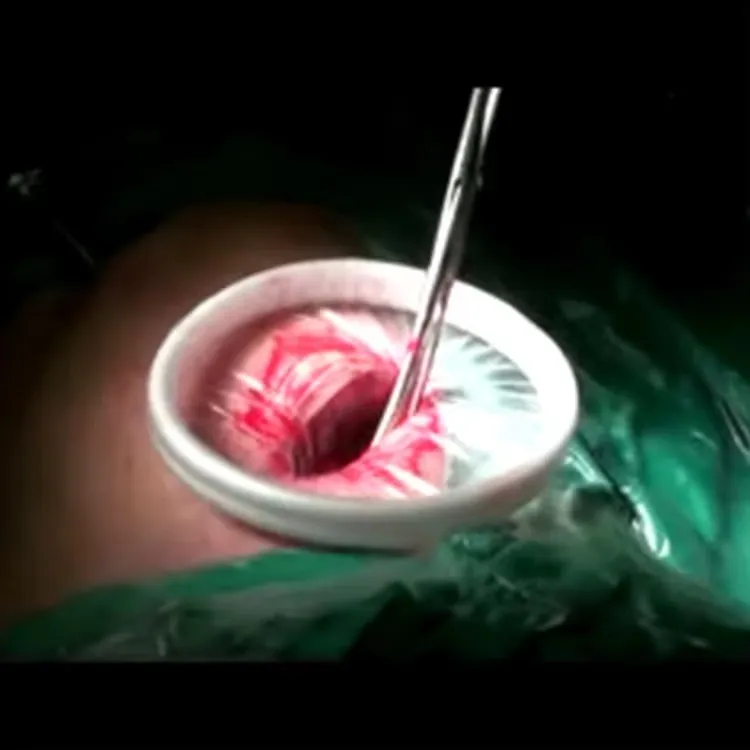
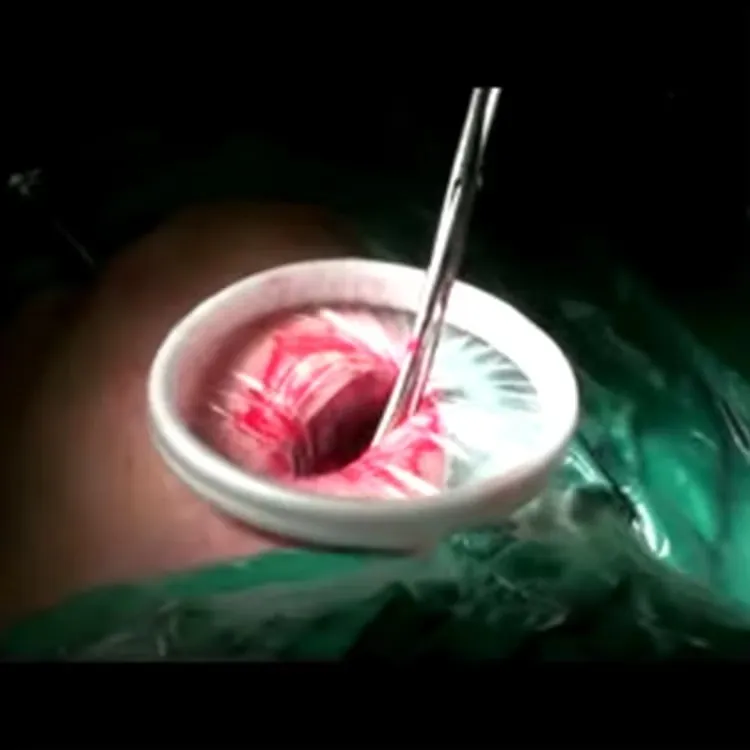
Wound Protectors/Retractors (Model B) Specification
| Product Code | Diameter (mm) | Incision Size (mm) |
|---|---|---|
| WP-40B | 40 | 15~25 |
| WP-60B | 60 | 25~35 |
| WP-90B | 90 | 35~50 |
| WP-120B | 120 | 45~70 |
| WP-150B | 150 | 60~90 |
| WP-180B | 180 | 75~105 |
| WP-200B | 200 | 80~120 |
| WP-230B | 230 | 90~130 |
| WP-250B | 250 | 100~140 |
| WP-280B | 280 | 120~160 |
| WP-330B | 330 | 150~180 |

