The global Surgical Instrument Sterilization Packaging market is undergoing a period of rapid evolution, driven by the increasing complexity of surgical procedures and the non-negotiable demand for patient safety. As healthcare systems worldwide grapple with hospital-acquired infections (HAIs), the role of the Sterile Barrier System (SBS) has transitioned from simple containment to a sophisticated engineering discipline. Today, the industry is valued at billions of dollars, with a steady growth trajectory fueled by aging populations in North America and Europe, alongside burgeoning healthcare infrastructure in the Asia-Pacific region.
Driven by an increase in elective surgeries and the rise of ambulatory surgery centers (ASCs), the demand for high-performance medical-grade paper and Tyvek® pouches is at an all-time high.
Strict adherence to ISO 11607 and the EU Medical Device Regulation (MDR) has become the gold standard, requiring factories to provide extensive validation for seal integrity and microbial barrier performance.
Modern procurement teams are increasingly seeking "green" alternatives—recyclable mono-materials and reduced-plastic solutions that do not compromise the sterile integrity of the device.
Dongguan LeienCare Tech Co., Ltd. is a professional manufacturer specializing in surgical instruments, disposable surgical supplies, and sterile medical products, serving global healthcare markets as a reliable OEM supplier. The company is committed to delivering high-quality, safe, and efficient medical solutions for hospitals, clinics, laboratories, and healthcare distributors worldwide.
LeienCare Tech offers a comprehensive product portfolio that includes a wide range of surgical instruments, operating room consumables, sterile packaging solutions, and disposable medical supplies designed to meet diverse clinical requirements. All products are manufactured under strict quality control systems and comply with international medical standards to ensure precision, reliability, and patient safety in critical medical environments.
Purchasing managers for large hospital groups and medical device manufacturers (MDMs) are no longer just looking for a "pouch." They are seeking Total Packaging Solutions that offer:
Central Sterile Supply Departments utilize our SMS Sterilization Wraps for large tray sets, ensuring heavy surgical kits remain sterile for long-term storage.
Small-scale dental practices rely on our Self-Seal Pouches for quick, reliable sterilization of drills and handpieces between patients.
Manufacturers of implants use our Custom Tyvek Blisters to provide a rigid, puncture-resistant barrier for high-value orthopedic devices.
Veterinary surgeons use our specialized TPLO Saw Sterilization Boxes to maintain the life and sterility of expensive power tools.
As a leading Surgical Instrument Sterilization Packaging Factory based in Dongguan, China, LeienCare Tech provides a strategic advantage to our global partners:
With precision manufacturing capabilities and a strong focus on hygiene, we maintain consistent product quality and scalable production capacity.
We support deep customization in product design, materials, packaging, and private labeling to meet specific customer and market needs.
Located in the heart of China's industrial hub, we offer streamlined logistics and competitive lead times for international distributors.
Driven by innovation, quality assurance, and customer satisfaction, LeienCare Tech continues to expand its global reach. The company is dedicated to supporting healthcare professionals with dependable surgical and disposable medical products that enhance operational efficiency and contribute to improved patient care outcomes. Our production line incorporates state-of-the-art automation to ensure that every seal is airtight and every indicator is accurate.



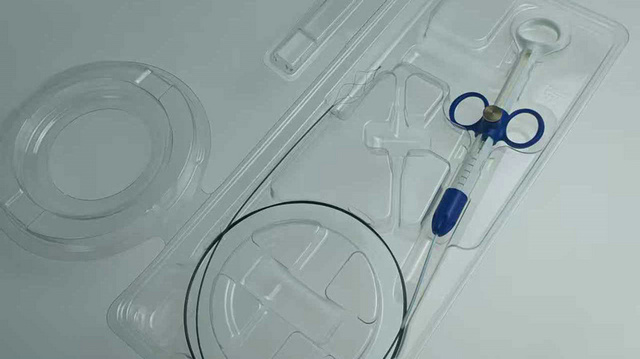




“Our mission is to bridge the gap between high-end medical technology and accessible healthcare solutions. By optimizing our manufacturing processes in Dongguan, we provide the world with surgical instrument packaging that meets the most rigorous clinical standards while remaining economically viable for hospitals everywhere.”