Company Profile
Established in 1958, we are a professional enterprise engaged in the development, manufacture, and sales of orthopedic implants and instruments. As a director company of the Medical Apparatus Industry Association, our product line covers spine pedical screws, interlocking nails, locking plates, trauma screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Equipped with advanced CNC machines and high-precision digital control technology, we operate within a 100,000 Grade axenic purification chamber to ensure the highest product quality. We are committed to "perfect design, innovative technology, and top-ranking products" to serve the global medical community.

Frequently Asked Questions
What are your primary orthopedic products?
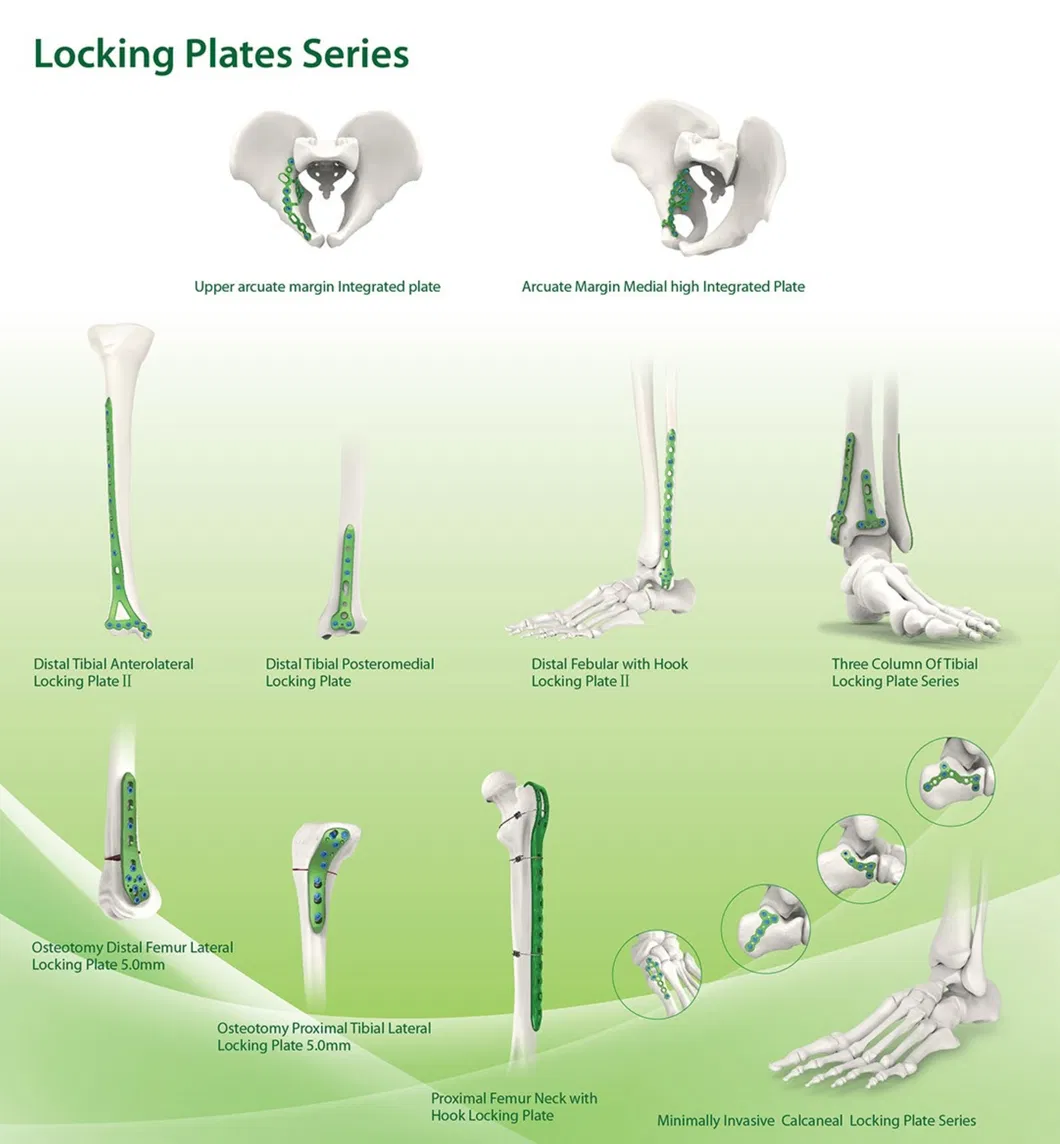
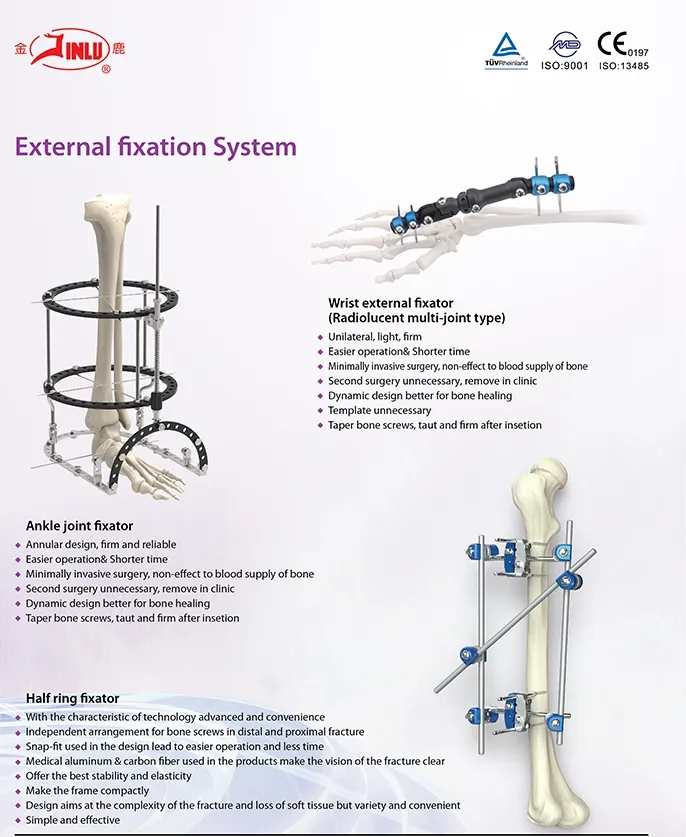
Our main products include metal bone plates and screw systems, cannulated bore screws, interlocking intramedullary nails, spinal fixation devices, and external fixation devices.
How do you ensure the quality of surgical instruments?
All products are manufactured according to EU CE standards. We conduct pre-production sample testing and a final rigorous inspection before every shipment.
Do you offer OEM services?
Yes, we provide OEM services and can customize products according to specific medical requirements and branding needs.
What certifications does your facility hold?
We are the first in our sector to pass ISO 9001 quality system certification, and our products hold CE, FDA, and ISO13485 approvals.
What are the available shipping and payment terms?
We accept various delivery terms including FOB, CIF, EXW, and Express Delivery. Payments can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
What is your monthly production capacity?
Our current production capacity is approximately 10,000 pieces per month, ensuring a stable supply for large-scale orders.