Basic Information
| Product Type |
Disposable Plastic Ampoule / Cartridge |
| Sterilization |
Ethylene Oxide (EO) Sterilization |
| Guarantee Period |
5 Years |
| Compatibility |
Needle-Free (Jet) Injector System |
| Capacity |
0.35 mL / 0.5 mL |
| Use |
Single-Use Only |
| Material |
Medical-grade plastic |
| OEM/ODM |
Available |
Product Introduction
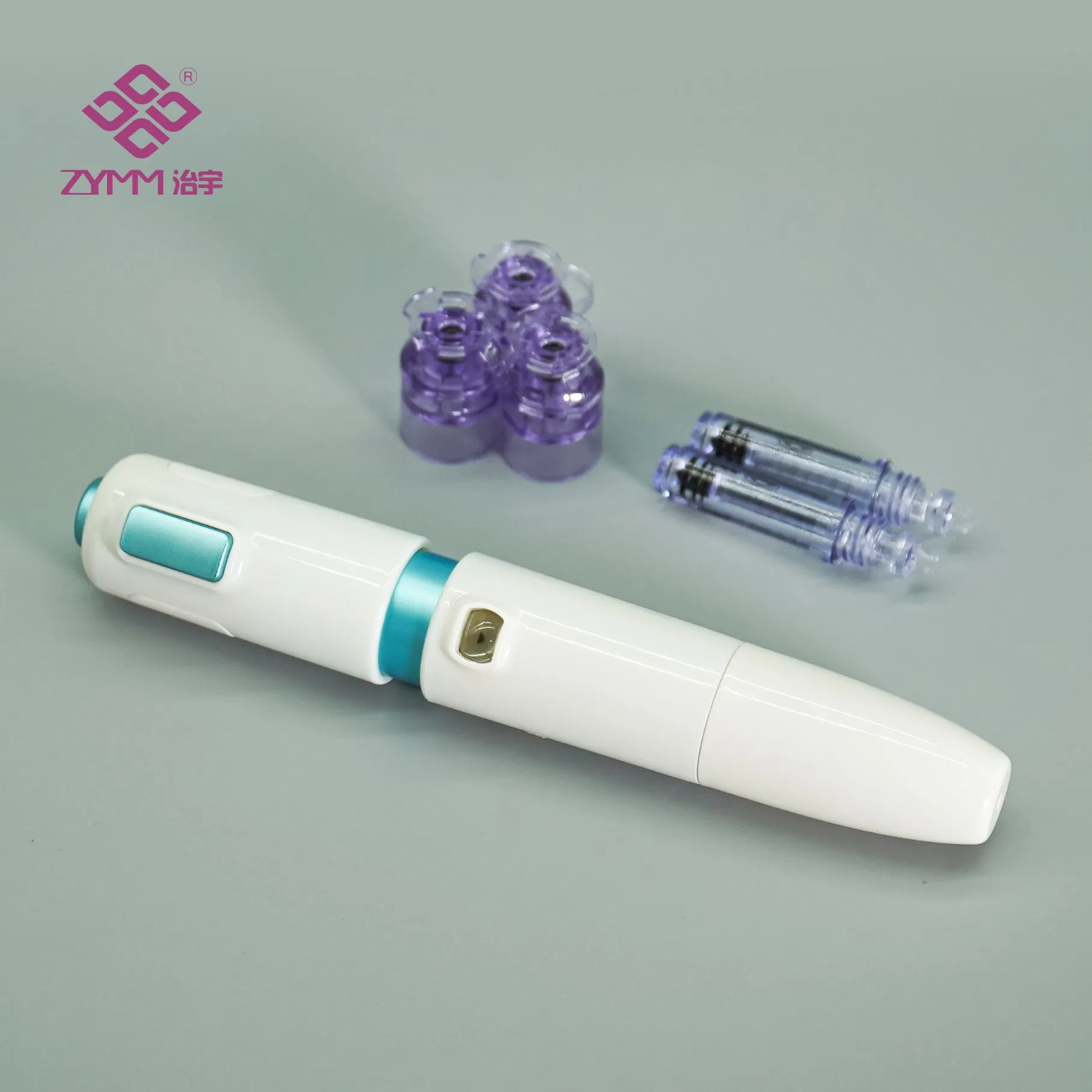
The Disposable Plastic Ampoule/Cartridge is a high-precision consumable designed specifically for needle-free (jet) injector systems. It serves as the primary container for liquid medication, facilitating subcutaneous or intradermal administration. The leak-resistant structure and transparent body allow for precise volume observation and safe delivery.
Transparent body for clear medication visibility
Leak-resistant design for dosage accuracy
Compatible with specialized filling devices
Multiple capacities: 0.35mL and 0.5mL
Main Product Display
Professional Manufacturing
Established in 2005, the manufacturer specializes in disposable sterile medical consumables. Operating within a large-scale facility that covers 32,000 m², the production site includes over 12,000 m² of ISO Class 8 (Class 100,000) cleanroom environments. With 8 EO sterilization cabinets and an in-house testing center for biological, chemical, and physical analysis, the facility ensures consistent quality for global supply.
The product portfolio includes high-volume medical consumables, medical aesthetic disposables, veterinary health products, dental components, and professional tattoo/piercing needles. Annual output exceeds 1 billion pieces for syringes, supported by advanced automation and rigorous QC inspection.
Company Advantages
Certifications & Compliance
Quality management systems are ISO 13485 certified. Compliance includes EU MDR 2017/745 QMS certification (TUV Rheinland), FDA establishment registration, and 510(k) clearance for specific product families. Complete market-specific documentation is available to support international distribution.
Global Exhibition
Packaging & Shipping
Flexible packaging options include PE pouches, blister packaging, and customized export-ready cartons. Shipments are typically arranged on FOB terms, with full support for artwork, private labeling, and multi-language instructions for use (IFU).
Frequently Asked Questions
Are you a manufacturer or a trading company?
We are a manufacturer specializing in single-use medical devices and sterile consumables since 2005.
Do you provide OEM/ODM services?
Yes. We support private labeling, customized packaging, artwork/printing, and multi-language labels/IFU.
What sterilization method is used for these products?
Ethylene Oxide (EO) sterilization is used for all sterile products, following strict internal QC and customer requirements.
Can you provide samples for evaluation?
Yes, samples are available. Shipping is arranged via courier; costs depend on the item and quantity requested.
What quality standards do you follow?
We operate an ISO 13485 certified Quality Management System and provide supporting documentation such as CE and FDA registration depending on the product group.
What is the typical lead time for orders?
Lead time depends on product type, quantity, and packaging. A specific production schedule is provided after specification and artwork confirmation.