The global laparoscopic instruments market is witnessing an unprecedented CAGR driven by the "Minimally Invasive First" approach. From intricate Maryland Dissectors to high-frequency bipolar graspers, the engineering required must meet sub-millimeter tolerances. Our focus at LeienCare Tech is to bridge the gap between complex surgical requirements and scalable manufacturing reality.
The shift from reusable to high-quality disposable laparoscopic tools is reducing cross-contamination risks and hospital overhead globally.
Leveraging medical-grade 304/316L stainless steel and biocompatible polymers to ensure structural integrity during high-torque procedures.
Developing instruments with integrated sensors for robotic-assisted platforms, anticipating the future of autonomous surgery.
Global procurement teams in the medical sector face unique challenges: regulatory fragmentation, supply chain volatility, and the "Information Gain" deficit. LeienCare Tech addresses these through a holistic OEM/ODM framework.
Dongguan LeienCare Tech Co., Ltd. is a professional manufacturer specializing in surgical instruments, disposable surgical supplies, and sterile medical products. The company is committed to delivering high-quality, safe, and efficient medical solutions for hospitals, clinics, laboratories, and healthcare distributors worldwide. Our macro solutions include:
The roadmap for laparoscopic tool development is anchored in three pillars: Miniaturization, Intelligence, and Ergonomics.
Phase 1: Precision Refinement (Present) - Enhancing haptic feedback in reusable graspers and needle holders. Ensuring Yasargil micro-needle holders provide the tactile sensitivity required for delicate sutures.
Phase 2: Hybrid Instruments - Combining bipolar energy with mechanical cutting in a single 5mm port tool, reducing instrument exchange times.
Phase 3: Smart-Sensing End-Effectors - Incorporating micro-sensors to monitor tissue pressure, preventing accidental trauma during retraction.
Navigating the transition from MDD to MDR (Medical Device Regulation) in Europe and meeting FDA 510(k) requirements in the US is the hallmark of a reliable supplier. LeienCare Tech maintains rigorous quality control systems that go beyond standard ISO requirements.



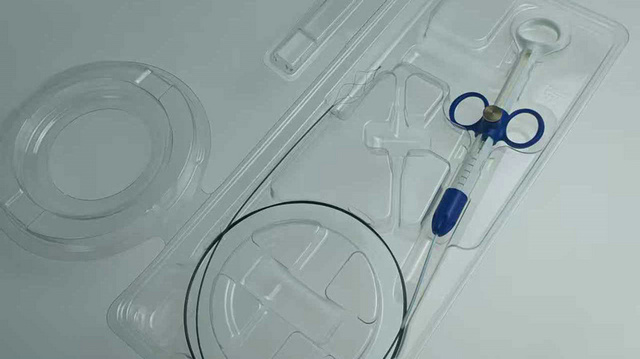
With advanced production facilities, precision manufacturing capabilities, and a strong focus on hygiene and sterilization processes, we maintain consistent product quality and scalable production capacity. Our flexible OEM and ODM services support customization in product design, materials, packaging, and private labeling.
A: We utilize medical-grade martensitic stainless steel for cutting edges (scissors) to maintain sharpness, and austenitic steel for handles and shafts for superior corrosion resistance during repeated sterilization cycles.
A: Our OEM workflow includes laser marking for logos, custom color-coding for handle identification, and bespoke sterile pouch design to meet regional regulatory standards.
A: We are currently exploring bio-based polymers and specialized medical waste recycling programs to help hospitals achieve their sustainability goals without compromising safety.
A: Typically, 4-6 weeks for initial CAD design and functional prototyping, followed by rigorous bench testing before moving to pilot production.



