
1 / 5










| Product Code | Product Name | Model | Description |
|---|---|---|---|
| 030020003 | Kyphoplasty Tool Kit | Type B Lumbar Spine |
φ3.0 Percutaneous Access Device 1pc, φ3.5 Aiguille 1pc, Guide Wire 1pc, φ4.2 Expansion Cannula 1pc, φ3.5 Bone Cement Applier 4pc |
| 030020002 | Kyphoplasty Tool Kit | Type D Lumbar Spine |
φ3.0 Percutaneous Access Device 2pc, φ3.5 Aiguille 1pc, Guide Wire 2pc, φ4.2 Expansion Cannula 2pc, φ3.5 Bone Cement Applier 6pc |
| 030020001 | Kyphoplasty Tool Kit | Type G Lumbar Spine |
φ4.2 Percutaneous Access Device 1pc, φ3.5 Aiguille 1pc, Guide Wire 1pc, φ3.5 Bone Cement Applier 4pc |
| 030030005 | Kyphoplasty Tool Kit | Type T1 Thoracic Spine |
φ2.4 Percutaneous Access Device 2pc, φ3.0 Aiguille 1pc, Guide Wire 2pc, φ3.6 Expansion Cannula 2pc, φ3.0 Bone Cement Applier 6pc |
| 030030006 | Kyphoplasty Tool Kit | Type T11 Thoracic Spine |
φ2.4 Percutaneous Access Device 1pc, φ3.0 Aiguille 1pc, Guide Wire 1pc, φ3.6 Expansion Cannula 1pc, φ3.0 Bone Cement Applier 4pc |
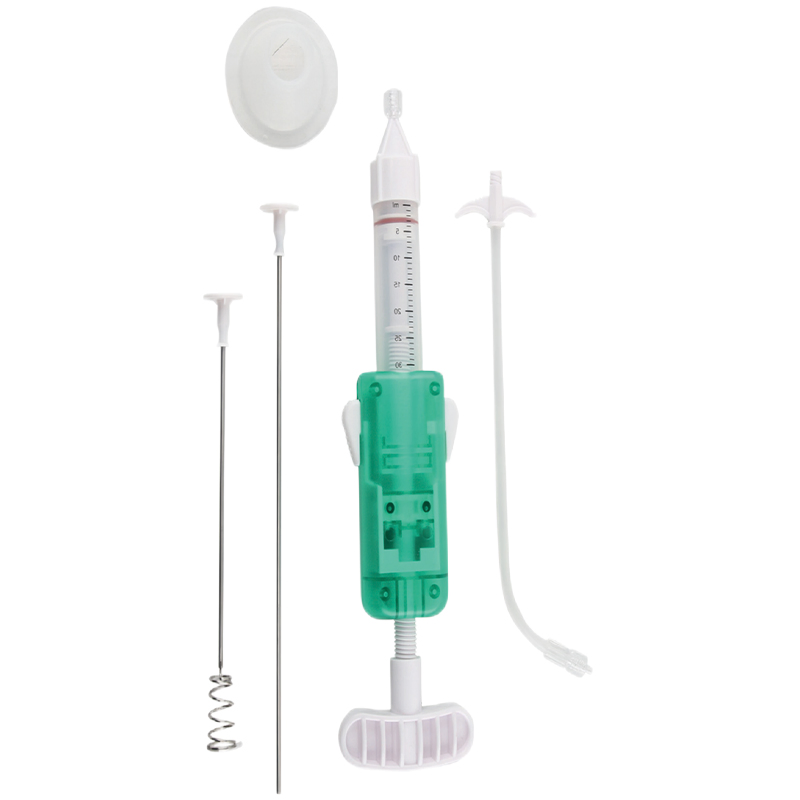
Integrated, one-step design for quick and efficient percutaneous access to bone and create bone tissue guide channel. Effectively reduces trauma during procedures. Available with bevel or diamond tips to suit clinical needs.
*All prices are for reference only. Exact price will be based on the latest quotation.