| Model NO. | Pure water equipment |
| Certification | CE, GMP, ISO |
| Control System | Fully Automatic |
| Treatment Process | EDI, Filtration, RO, Softening, Ultrafiltration, UV Disinfection |
| Material | 304 Stainless Steel |
| Precision | High Precision |
| Membranes Brand | Dow etc |
| Usage | Pure Water Production |
| Output Capacity | Custom |
| HS Code | 842121 |
| Production Capacity | 2000sets Per Year |

Premium distilled WFI Water Treatment System for Injection (WFI)
Our machine lines comply with international standards including European pharmacopoeia EP, US pharmacopoeia USP, and adhere to WHO GMP, EU cGMP, and US FDA regulations.
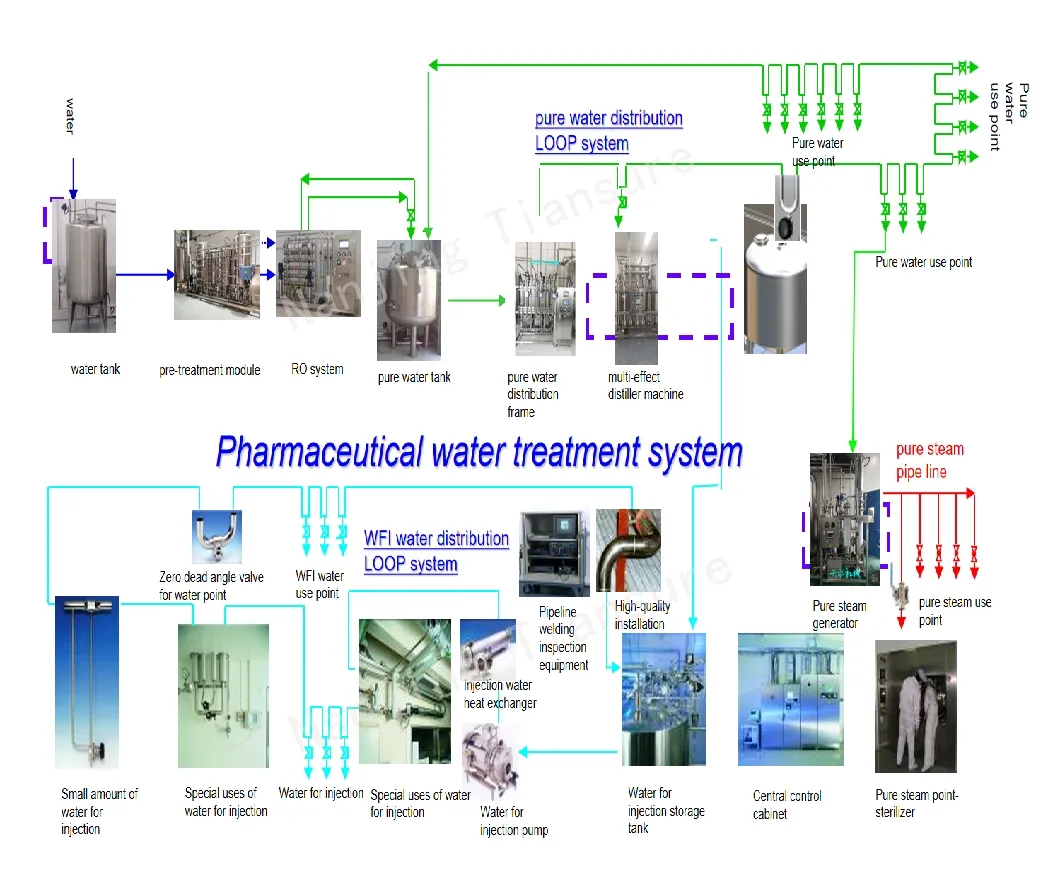




Incorporating advanced production technology for multi-effect distilled water machines, these systems represent world-class standards. Available in capacities from 300-5000, control modes range from manual to fully automatic PLC systems.
Features: Double tube sheets prevent cross-contamination. Flanged sealing structures prevent leakage. High-efficiency heat exchange saves 20% energy and significantly reduces cooling water consumption.
Design pressure reaches 0.88Mpa, ensuring a long service life and high water output. Our energy-saving technologies provide significant operational advantages.
Design Standards:
• cGMP
• ISO 9001:2000
• European Pressure Equipment Directive 97/23/EC
• ASME BPE
• IEC 60204-1


We specialize in providing energy-efficient pharmaceutical water and liquid preparation solutions. Our expertise serves biopharmaceuticals, medical devices, aesthetics, and electronics industries across global markets.
Certified with ISO9001, CE, and GC2 special equipment installation qualifications. All production complies with strict GMP standards for safety and reliability.
Utilizing modular design and 316L stainless steel for high corrosion resistance. Automated controls improve efficiency while providing customized process requirements.
Extensive history serving pharmaceutical and medical device leaders worldwide, demonstrating high market trust through large-scale project execution.
24-hour rapid response maintenance, technical training, and full assistance with GMP certification and documentation (URS, DQ, IQ, OQ, PQ).